| Names | |
|---|---|
| IUPAC name | |
| Other names Nickel hydroxide, Theophrastite | |
| Identifiers | |
| |
| ChemSpider | |
| ECHA InfoCard | 100.031.813 |
| EC Number |
|
| RTECS number | |
CompTox Dashboard(EPA) | |
| |
| |
| Properties | |
| Ni(OH)2 | |
| Molar mass | 92.724 g/mol (anhydrous) 110.72 g/mol (monohydrate) |
| Appearance | green crystals |
| Density | 4.10 g/cm3 |
| Melting point | 230 °C (446 °F; 503 K) (anhydrous, decomposes) |
| 0.13 g/L | |
| +4500.0·10−6 cm3/mol | |
| Structure[1] | |
| hexagonal, hP3 | |
| P3m1, No. 164 | |
α = 90°, β = 90°, γ = 120° | |
| Thermochemistry | |
| 79 J·mol−1·K−1[2] | |
Std enthalpy of formation(ΔfH⦵298) | −538 kJ·mol−1[2] |
| Hazards | |
| Safety data sheet | External SDS |
| GHS pictograms | [3] |
| GHS Signal word | Danger[3] |
| H302, H332, H315, H334, H317, H341, H350, H360, H372[3] | |
| P260, P284, P201, P280, P405, P501[3] | |
| Lethal dose or concentration (LD, LC): | |
| 1515 mg/kg (oral, rat) | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| verify (what is ?) | |
| Infobox references | |
The formula causes a massive difference between a medium and a large reduction: 75% reduction (¼ original armor) is essentially twice more than 50% reduction (½ original armor). Because of the twofold reduction, a simple (1 + HM ) × (1 + AM ) calculation yields incorrect results. The chemical properties of THC vs CBD. As with many of the cannabinoids, THC and CBD have low solubility in water, but good solubility in most organic solvents, particularly lipids and alcohols. Both THC and CBD are present in cannabis in a mixture of acidic forms, which are readily de-carboxylated and chemically altered upon heating, important when you consider that smoking cannabis is the most common form of consumption.
The test tube in the middle contains a precipitate of nickel(II) hydroxide
Nickel(II) hydroxide is the inorganic compound with the formula Ni(OH)2. It is an apple-green solid that dissolves with decomposition in ammonia and amines and is attacked by acids. It is electroactive, being converted to the Ni(III) oxy-hydroxide, leading to widespread applications in rechargeable batteries.[4]
- Confirmatory Tests For Cations. Confirmatory Tests For Cations. 31 Questions. What is the chemical formula of DMG. Chemical formula of aluminon reagent. Chemical formula of ammonium sulfite. Chemical formula of barium nitrate. Chemical formula of ammonium aurin-tricarboxylate. Chemical formula of Lead(II) Nitrate.
- Of air is included. The formula allows for the determination of the weight in vacuum, W after correction, where W a is the weight in air. The volume is calculated from the weight and the density. W = W a + 1.06 W a /1000 Table. Density of water at various temperatures. Temperature (°C ) Density of water ( g mL-1) 26 0.99681 27 0.99654 28 0.99626.
Properties[edit]
Nickel(II) hydroxide has two well-characterized polymorphs, α and β. The α structure consists of Ni(OH)2 layers with intercalated anions or water.[5][6] The β form adopts a hexagonal close-packed structure of Ni2+ and OH− ions.[5][6] In the presence of water, the α polymorph typically recrystallizes to the β form.[5][7] In addition to the α and β polymorphs, several γ nickel hydroxides have been described, distinguished by crystal structures with much larger inter-sheet distances.[5]
The mineral form of Ni(OH)2, theophrastite, was first identified in the Vermion region of northern Greece, in 1980. It is found naturally as a translucent emerald-green crystal formed in thin sheets near the boundaries of idocrase or chlorite crystals.[8] A nickel-magnesium variant of the mineral, (Ni,Mg)(OH)2 had been previously discovered at Hagdale on the island of Unst in Scotland.[9]
Reactions[edit]
Dmg Chemical Formula
Nickel(II) hydroxide is frequently used in electrical car batteries.[6] Specifically, Ni(OH)2 readily oxidizes to nickel oxyhydroxide, NiOOH, in combination with a reduction reaction, often of a metal hydride (reaction 1 and 2).[10]
Reaction 1 Ni(OH)2 + OH− → NiO(OH) + H2O + e−
Reaction 2 M + H2O + e− → MH + OH−
Net Reaction (in H2O)Ni(OH)2 + M → NiOOH + MH
Of the two polymorphs, α-Ni(OH)2 has a higher theoretical capacity and thus is generally considered to be preferable in electrochemical applications. However, it transforms to β-Ni(OH)2 in alkaline solutions, leading to many investigations into the possibility of stabilized α-Ni(OH)2 electrodes for industrial applications.[7]
Synthesis[edit]
The synthesis entails treating aqueous solutions of nickel(II) salts with potassium hydroxide.[11]
Toxicity[edit]
The Ni2+ ion is a known carcinogen. Toxicity and related safety concerns have driven research into increasing the energy density of Ni(OH)2 electrodes, such as the addition of calcium or cobalt hydroxides.[4]
See also[edit]
Dmg Chemical Formula Chart
References[edit]
- ^Enoki, Toshiaki; Tsujikawa, Ikuji (1975). 'Magnetic Behaviours of a Random Magnet, NipMg(1-p)(OH2)'. Journal of the Physical Society of Japan. 39 (2): 317. doi:10.1143/JPSJ.39.317.
- ^ abZumdahl, Steven S. (2009). Chemical Principles 6th Ed. Houghton Mifflin Company. p. A22. ISBN978-0-618-94690-7.
- ^ abcd'Nickel Hydroxide'. American Elements. Retrieved 2018-08-30.
- ^ abChen, J.; Bradhurst, D.H.; Dou, S.X.; Liu, H.K. (1999). 'Nickel Hydroxide as an Active Material for the Positive Electrode in Rechargeable Alkaline Batteries'. J. Electrochem. Soc. 146 (10): 3606–3612. doi:10.1149/1.1392522.
- ^ abcdOliva, P.; Leonardi, J.; Laurent, J.F. (1982). 'Review of the structure and the electrochemistry of nickel hydroxides and oxy-hydroxides'. Journal of Power Sources. 8 (2): 229–255. doi:10.1016/0378-7753(82)80057-8.
- ^ abcJeevanandam, P.; Koltypin, Y.; Gedanken, A. (2001). 'Synthesis of Nanosized α-Nickel Hydroxide by a Sonochemical Method'. Nano Letters. 1 (5): 263–266. doi:10.1021/nl010003p.
- ^ abShukla, A.K.; Kumar, V.G.; Munichandriah, N. (1994). 'Stabilized α-Ni(OH)2 as Electrode Material for Alkaline Secondary Cells'. J. Electrochem. Soc. 141 (11): 2956–2959. doi:10.1149/1.2059264.
- ^Marcopoulos, T.; Economou, M. (1980). 'Theophrastite, Ni(OH)2, a new mineral from northern Greece'(PDF). American Mineralogist. 66: 1020–1021.
- ^Livingston, A.; Bish, D. L. (1982). 'On the new mineral theophrastite, a nickel hydroxide, from Unst, Shetland, Scotland'(PDF). Mineralogical Magazine. 46 (338): 1. doi:10.1180/minmag.1982.046.338.01.
- ^Ovshinsky, S.R.; Fetcenko, M.A.; Ross, J. (1993). 'A nickel metal hydride battery for electric vehicles'. Science. 260 (5105): 176–181. doi:10.1126/science.260.5105.176. PMID17807176.
- ^Glemser, O. (1963) 'Nickel(II) Hydroxide' in 'Handbook of Preparative Inorganic Chemistry, 2nd ed. G. Brauer (ed.), Academic Press, NY. Vol. 1. p. 1549.
External links[edit]
Retrieved from 'https://en.wikipedia.org/w/index.php?title=Nickel(II)_hydroxide&oldid=904764643'
31 Questions | By Antonsison16 | Last updated: Feb 23, 2014
H-dmg Chemical Formula
Settings
Success! A copy of this quiz is in your dashboard.
Yeah. Based on the cheat sheet + stuff ma'am gave us. Yeah. Does not include much of the proceduresstudy day 4-5 on your own nalang haha.
- ____ is added to Copper to confirm its presence, yielding a blue color.
- The color of Mg^2+ in the final step of its confirmatory test is ___.
- Red
- Blue
- Colorless
- Green
- HNO3 is added to destroy/decompose the ammine complex, and makes AgCl repricipitate.
- True
- False
- Unknown + 2OH^- → white precipitatewhat could the unknown be?(2 answers)
- Ag +
- Fe 3+
- Ni 2+
- Zn 2+
- Cu 2+
- Ca 2+
- Mg 2+
- Dmg file extractor. Al 3+
- ___ is added in the final step of Ni^2+ to confirm it, yielding a bright red color.
- Ag^+ + HCl → ___?
- Cu^2+ + NH3 → ???
- CH3COOH is added to make the solution slightly ____ for Ca^2+
- Questions 9 - 12 will be for the confirmatory test of Ag^+.Ag^+ + ___ will yield ___(2 pts)
- 3OH -
- 2OH
- HCl
- Ammonia
- AgOH
- AgCl
- Ag +
- (answer in # 9) + ___ will yield ___ + ____.(3 pts)
- Ammonia
- Ag[NH3]2 +
- Cl -
- HNO3
- Ag +
- 2NH3
- 3OH -
- (answer in #10) + HNO3 will yield ____ + ____
- AgCl (s) + 2NH4 +
- Ag + + Cl -
- 2NH3 + 2H +
- Ag + + 2NH3
- which 2 equations will yield the final result of[Ag(NH3)2] + + Cl - + 2H + yielding AgCl (s) + 2NH4 +(aq)(white, crystalline)
- 2NH3 + 2H^+ yielding 2NH4^+
- 2NH3 + 2OH- yielding N2O2 + H8
- Ag^+ + Cl^- yielding AgCl (s)
- Ag^+ + HCl yielding AgCl (s)
- The color of Fe 3+ is(according to Ma'am)
- Brown
- Orange
- Red brown
- Dark red
- The first step of Ni^2+Ni^2+ + 3OH^- yielding Ni(OH)3(s) will be color
- Red
- Bright red
- White
- Colorless
- None of the above
- The first reagent added to Zn^2+ is_____.
- NaOH
- OH^-
- NOH
- H+
- To confirm Cu^2+ the solution must be
- Acidic
- Basic
- Which Precipitation reactions yield no reactions(6 answers)
- Na2SO4 & Ba(NO3)2
- FeCl3 & Ba(NO3)2
- Pb(NO3)2 & Ba(NO3)2
- KSCN & Ba(NO3)2
- K2Cr2O7 & Ba(NO3)2 Download amazon prime video to mac.
- K2CrO4 & Ba(NO3)2
- FeCl3 & Na2SO4
- Pb(NO3)2 & Na2SO4
- KSCN & Na2SO4
- K2Cr2O7 & Na2SO4
- K2CrO4 & Na2SO4
- Pb(NO3)2 & FeCl3
- KSCN & FeCl3
- K2Cr2O7 & FeCl3
- K2CrO4 & FeCl3
- KSCN & Pb(NO3)2
- K2Cr2O7 & Pb(NO3)2
- K2CrO4 & Pb(NO3)2
- K2Cr2O7 & KSCN
- K2CrO4 & KSCN
- K2CrO4 & K2Cr2O7
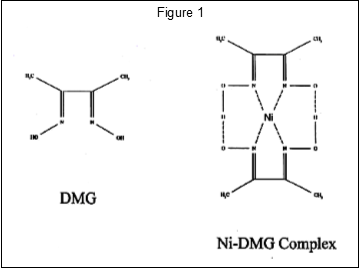
- What is the chemical formula of DMG
- Chemical formula of aluminon reagent
- Chemical formula of potassium dichromate
- Chemical formula of thioacetamide
- Chemical formula of ammonium sulfite
- Chemical formula of barium nitrate
- Chemical formula of ammonium aurin-tricarboxylate
- Chemical formula of Lead(II) Nitrate
- Chemical formula of Potassium thiocyanate
